If conversations between you and your company's head of quality typically revolve exclusively around headcount, departmental expenditures and other belt-tightening measures, you may be missing out on cost-effective ways to both streamline your manufacturing process and make your company more profitable.
Let me give you just one example, based on the expertise of my company, Celsis International Ltd., around rapid microbial testing. It's science, yes, and perhaps not your area of expertise. But don't assume that your company's micro lab is going to come to you with a proposal to increase its budget, even if it does lower overall business costs. Remember, their marching orders include keeping their expenses low.
The problem is that, since the costs of adopting a rapid method fall under the Quality department, yet the savings accrue to Manufacturing, it's not easy for individual departments to see beyond their budgets to the larger – and readily achievable -- opportunity of saving the company time and money every day, with every production run. Let's look briefly at the source of the savings.
A Lean Solution
In Lean manufacturing terms, saving days in the production cycle translates into significant quantifiable savings in reduced inventory requirements, shortened lead time and faster recovery from contamination events. The steps on the graph in Figure 1 show value-added activities.
In the red section, the plateaus represent micro testing hold times. The first is for raw materials testing, the second is in-process screening and the top horizontal line represents hold time for finished product testing. For each area where micro-hold is applied, three to seven days are required to detect contamination using traditional microbial limits methods; sterility testing adds 14-21 days.
This time is expensive: it significantly impacts manufacturing efficiency, responsiveness and working capital requirements, all of which ultimately affect your company's profitability and bottom line.
As you can see in the chart, adopting a rapid microbial method reduces the number of days needed to complete testing. The company in this example was able reduce its total cumulative inventory investment at any given moment in time by approximately $650,000.
Spending to Save
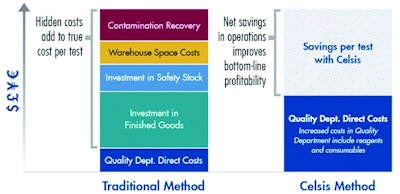
If your Quality department is concerned about adding rapid methods to their budget, they may not understand the value of a high-throughput screening system. It is true that the Quality department will have to increase its budget for a rapid system, but it will save your company much more. (See Figure 2.) It often takes someone looking at the larger, facility-wide financial picture to see the opportunity, and you can help open Quality's eyes to the bigger benefits available.
And, of course, it's important for you to be able to quantify all the benefits. The savings from rapid methods outlined above do not even include the many environmental benefits of a rapid method, including reductions in waste and in water and energy consumption. Some rapid system manufacturers offer tools, such as the financial and environmental impact assessments available from Celsis, which enable companies to estimate the value of adopting a rapid system using readily available data. This is the bigger picture that you need to present both to Quality and to senior management to win buy-in for the necessary investments in a new solution.
Start the Conversation
The point is, you must resolve now to engage your Quality department leaders in a discussion about innovative ways to reduce expenses, such as through the adoption of rapid methods. Once Quality (and senior management) understand that the company-wide benefits (e.g., of faster release time) far outweigh the incremental costs (e.g., of a rapid system), you'll be able to work together to find the right solution to deliver bottom-line benefits to the company as a whole. So let's get the conversation started today!
About the Author
Cindy Lieberman is vice president with Celsis International Ltd. She can be reached at [email protected].