There are few industries as tightly interwoven with life and death issues as medical equipment. Getting everything right—from adherence to regulations to on-time product deliveries to perfectly performing products—is paramount. In the medical equipment supply chain, complete transparency is an absolute necessity. Transparency cannot be a part-time or a partial commitment. It must be an all-out endeavor to help achieve safer and more effective patient healthcare.
The regulatory pressure in the medical equipment industry is extraordinarily intense. Manufacturers of medical products—such as sonograms and X-ray machines—must comply with strict regulations and laws that govern safety and performance. Inaccurate product information in the medical equipment supply chain can threaten public health or induce penalties that either jeopardize companies’ abilities to operate or even put them out of business.
To help avoid these scenarios, it’s critical that medical equipment technology companies create end-to-end supply chain visibility that streamlines and accelerates product flows and increases awareness for all supply chain participants. Leveraging this framework, product delays, shortages and overstocks can be resolved before causing significant problems.
Medical supply chain objectives
The primary objective of medical supply chain visibility is to leverage increased transparency of product-related information to enable smoother and more efficient end-to-end processes; and effective regulatory compliance and improved decision-making—while avoiding redundancies, reconciliations and rework. Medical supply chain visibility consists of the complete management of product-related information along the supply chain from suppliers and original equipment manufacturers (OEMs) to customers in various countries. Information that needs to be managed includes master data; materials; products; bills of material (BOMs); vendors; customers; and transactional data such as purchase orders, manufacturing orders, delivery notes and invoices.
Medical supply chain visibility can deliver a range of potential benefits for medical equipment technology (MET) companies including cost savings, faster growth and reduced trade compliance risks. These companies face specific challenges in regulatory compliance.
The safety-critical nature of medical devices; the importance of regulatory compliance and tracing; and monitoring and controlling processes and data flows throughout the supply chain—combined, these elements create an even stronger need for supply chain visibility.
Supply chain visibility enhances patient safety and reduces regulatory compliance risks by maintaining historical tracking of device changes; delivering current and correct compliance data; and providing accurate and timely information of pre- and post-market approvals throughout the supply chain.
Improved regulatory compliance capabilities also help boost revenues and reduce costs. These potential benefits can be achieved by leveraging better re-use of submission information for different countries and products, which shortens ‘submission to approve’ cycle times; and enables the consistent use of globally harmonized processes, standards and templates.
Supply chain visibility can bolster revenues in other ways including greater process automation; more rapid data exchange; and faster cycle times throughout the supply chain. As organizations in other industries already learned, information transparency results in improved operational and sales effectiveness; better and faster decision-making; more effective performance management; and lower costs across end-to-end supply chain operations. This transparency reduces manual intervention and rework in the order flow through improved data quality, accuracy, availability and further process automation especially in importing and exporting transactions.
‘Vitals’ for medical technology companies
Medical equipment companies face increasing price and cost pressures. Hospitals pooled their negotiating power to drive more competitive pricing while public spending cuts have become endemic. Competition grows from emerging market medical equipment manufacturers leveraging their lower cost bases to challenge established brands.
What’s more, with pervasive global economic volatility, customers in emerging and mature markets focus heavily on costs. Further price pressures stem from cost-cutting healthcare reforms now being considered or implemented in many countries. And even though U.S. healthcare reform intends to bring more people into insurance plans and increase projected national medical spending, the country’s high budget deficit may further intensify pressure for cost savings in the healthcare sector. In Germany, healthcare budgets are under less pressure due to the country’s stable economic environment. But other risk factors—such as the euro crisis and slowdown in global growth—may move healthcare reforms back onto the agenda.
Approach the issues
Superior supply chain visibility capabilities can drive high performance—a view grounded in Accenture’s medical supply chain visibility approach. As such, management and transparency of supply chain product-related information is fundamental to business success in all industry sectors. This is especially true for companies in the medical equipment industry, which is characterized by intensifying cost and price pressures and extensive regulation. Medical supply chain visibility permeates the whole enterprise and directly/or indirectly impacts most processes throughout an organization.
A phased approach can help achieve effective medical supply chain visibility—starting with a sharply defined roadmap that assesses a company’s current supply chain; and pinpoints steps needed to help achieve full visibility. Once that roadmap is developed, companies should consider shifting to an implementation stage involving organizations and people; processes; data; and technology.
We examine those four phases in more detail below.
Organizations and people—Companies need a defined framework that outlines supply chain governance. The framework should delineate supply chain roles and responsibilities such as training, organizational planning and change management. This framework needs to include gatekeepers who shepherd product release information throughout the medical supply chain.
Processes—Firms should establish definitive and enforceable governance and data management processes that determine how medical supply chain data travels. To make it easier to identify and correct problems as they occur, these processes should cover the entire ecosystem of manufacturers, suppliers, regulators and customers. Most processes should be built around product releases and regulatory compliance.
Data—To facilitate accurate dashboard and supply chain reports, data structures and guidelines pertaining to product release information should be established. One crucial structure is master data management (MDM), which is a set of disciplines and processes for driving accurate, complete, timely, and consistent data classification. MDM can accelerate data exchange within corporations and to third parties; reduce data duplication; expedite product deliveries; and reduce information delivery costs.
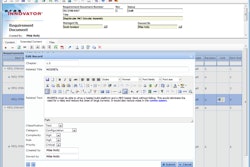
Technology—Medical companies should establish an IT landscape that supports supply chain processes. For example, they need to set up product data management and document management systems that store, run and access product-related information and product submission approvals.
For more information on the medical industry, download a copy of Accenture’s new report, “Supply Chain Visibility in the Medical Equipment Technology Industry.”